News
Although speakers at Battery Innovation Days this year underlined the delay of the first batches of end-of-life [EoL] electric vehicle batteries, mainly due to their extended life duration, participants unanimously agreed that recycling processes must evolve. This message comes in anticipation of the first massive return of EoL batteries. Traditional pyrometallurgical and hydrometallurgical processes, while effective for metal recovery, often overlook the non-metallic components: electrolytes, binders, and separators, thus missing out on a veritable circular economy.
The RHINOCEROS project has now validated several pre-treatment technologies at laboratory scale to recover valuable components, from the black mass, electrolyte solvents, binders, separators and current collectors (Cu, Al) from LIBs. In fact, in 2025, RHINOCEROS partners validated an integrated recycling process with a minimum efficiency of >90% for electrolyte, >80% for polymers and >95% for Li.
Over the past year, partners have tested and compared various pre-treatment processes, including mechanical shredding, vacuum drying, pyrolysis, supercritical CO₂ (scCO₂) extraction. Rigorous analysis reported two approaches that yield most promising results and later validated them at lab scale:
- Mechanical treatment combined with scCO₂ extraction for electrolyte and binder recovery: this technological route yields high material recovery – 99% electrolyte and 60% PVDF binder, with over 98% polymer purity. Using CO₂ as a recyclable solvent eliminates hazardous emissions and reduces energy demand. The process also improves workplace safety and produces cleaner black mass for downstream hydrometallurgical steps.
Learn more about scCO₂
- Thermal treatment [vacuum drying combined with pyrolysis] for organic removal and black mass preparation: a process technologically mature, already compatible with existing infrastructures, which delivered high-quality organic-free black mass and over 95% recovery of active materials. On the other hand, it remains an energy-intensive process that emits hazardous gases.
- Reactive milling, while technically interesting for cathode material reduction, was excluded due to its low technology readiness, slow kinetics in graphite-rich materials for mechanical route, and redundancy with high-temperature pathways.
The pre-treatment technologies studied during the research phase were combined into several routes that were candidates for upscaling. These have been evaluated by a series of criteria to choose the most appropriate technologies for the upscaling phase.
| Criteria | Route 2: Mechanical + scCO₂ | Route 4: Thermal Treatment |
| Technology Readiness | Medium – scCO₂ integration maturing | High – widely used in waste processing |
| Environmental Impact | Low energy use, minimal emissions, reusable CO₂ solvent | High energy demand, toxic gas emissions |
| CAPEX/OPEX | Moderate – scCO₂ adds cost but offsets via recovery | High – energy-intensive, complex gas handling |
| Material Recovery | High electrolyte & polymer recovery, moderate PVDF | Moderate electrolyte recovery (43%), no PVDF and partial fluorine recovery. |
| Black Mass Quality | Organic-free, high purity, suitable for leaching | Organic-free, reduced metals, ideal for leaching |
| Workplace Safety | Safer, fewer emissions | Fire hazard, HF/POF₃ gas risks, thermal control |
| Regulatory Compliance | Fewer regulatory hurdles for emissions; polymer recovery supports compliance | Established permitting pathway for pyrolysis but more complex exhaust control |
| Engineering Modelling | Scalable modular design, well-defined equipment chain, adaptable | Mature/established pyrolysis design, but complex off-gas treatment integration. |
As a conclusion, the two routes presented in the table were selected for upscaling. The first one combines mechanical process and supercritical CO2 technology for the solvent, polymer and fluorinated compounds recovery, while the second one is an optimised thermal pre-treatment for black mass production at pilot scale and electrolyte recovery.
Part of a joint dissemination initiative, RHINOCEROS, along with other members of the Cluster Hub “Materials for Batteries“, took the stage at Battery Innovation Days 2025, contributing to the discussion about the strategic role of recycling in the EU Battery Regulation and the broader circular economy.
Presentations explained, one after the other, Europe’s alternatives to build a circular battery ecosystem. With electric mobility accelerating and battery demand soaring, recycling is no longer optional. It has become a strategic necessity for Europe’s competitiveness and climate goals. And the EU wasted no time to announce on 3 December its recent ReSourceEU Action Plan, under the headline “Accelerating our critical raw materials strategy to adapt to a new reality”. ReSourceEU places circularity at the core of EU’s approach to set the basis for competitive CRMs industry in Europe. Projects similar to RHINOCEROS welcome this initiative and are willing to contribute with expertise and knowledge sourced from R&I.
Take-aways from the BID 2025 sessions
Eleonora Cali (RINA), representing the Materials for Batteries Cluster Hub in the parallel session “End of life, start of supply: Advancing battery recycling in Europe” on 2 December, joined leading experts to address two pressing realities in the battery industry:
- Europe’s dependency on imported raw materials. Lithium, cobalt, nickel, manganese and graphite are critical for the energy transition, yet supply chains remain dominated by non-European players. Recycling offers a way to keep these resources in Europe, reduce environmental impact, and comply with EU regulations on secondary raw material content. The European Commission’s new Battery Regulation aims to change that by mandating minimum recycled content for key materials from 2031. This is more than an environmental measure: it is an industrial policy designed to keep resources within Europe and reduce strategic dependency.
- the expected surge of end-of-life batteries. With EU speeding up its transition to electric mobility, the question of what happens to millions of batteries at the end of their lifetime is shifting from technical to strategic priority.
Surprisingly, speakers underlined EU’s anticipated timeline to develop recycling plants, with a scarce input of end of life applications. According to Andreas Opelt (Saubermacher) and Verena Fuchs (Cylib), for electric vehicle batteries, the timeline for returns is uncertain; early fleets are lasting longer than expected, delaying the recycling ramp-up. Opelt concluded his presentation with a pragmatic message: “The storm of batteries is coming, but if you build capacity too early, plants will sit empty”, arguing timing is critical.
Speakers from both R&I and industry taking the stage in this session called for:
- accelerated permitting for recycling infrastructures. In China, you can build a recycling plant in six months. In Europe, six months is not even enough to submit a permit,” Opel warned.
- enforcement of design-for-recycling standards in new battery regulations.
- support for industrial scale-up through funding but also simplified regulation.
- call to impose all possible measures to prevent black mass from exiting Europe, already reinforced by its recent classification as hazardous waste.
Probably one of the messages we take with us and integrate it to our initiatives’ objectives is that policies like the EU’s Critical Raw Materials Act, Battery Regulation and now recently adopted ReSourceEU provide the framework. What is needed now is execution at speed.

Production of raw materials for batteries from European resources
After Wednesday’s sessions on the EU political agenda, which outlined strategic measures to meet industry needs, attention shifted to the annual workshop of the Materials for batteries hub. Now in its 4th edition, the event was co-organised by Horizon Europe projects RELiEF, FREE4LIB, RESPECT and LITHOS. The focus of the workshop, true to its eponymous theme, was tackling one of Europe’s most urgent challenges: securing sustainable raw materials for batteries.
Under the inauguration of Oliver Schenk, Member of the European Parliament, this edition unfolded under the auspices of urgency, regulatory clarity and cross-border collaboration. The MEP called for swift implementation of the Critical Raw Materials Act and the Net Zero Industry Act, stressing the need for rapid permitting and the mobilisation of both public and private investment. “We cannot afford delays,” he warned. “This is about sovereignty.” His remarks were followed by strong appeals for cooperation among mining regions, manufacturing clusters, research centres and recycling hubs to build a resilient European value chain. He urged participants to contribute to upcoming legislative files, including the European Chips Act 2, the Circular Economy Act and the new EU budget, ensuring that the priorities of the battery materials community are embedded in future policies.
Nader Akil, founder of the Cluster Hub and moderator of the first technical session, emphasised on enhancing the dialogue between academia, industry and policy makers in order to ensure that these goals would be reached.
Following up with a presentation of his most recent scientific publication – “Lindy Effect in Hydrometallurgy” [co-authored with Dr. Ir. Peter Tom Jones] – Professor Koen Binnemans provided a frank look at the shortcomings in industrialising battery material innovations. Transferring hydrometallurgical advancements from lab to plant is slow, constrained by economics, regulation and what the authors call “the Lindy effect” – the tendency for established technologies to stand the test of time. Industry tends to favour incremental improvements to existing processes, such as reducing reagent consumption or increasing automation, rather than adopting entirely new chemistries, due to the high risks and costs associated with large-scale change. Launching a debating topic, audience questions shifted to technical and permitting challenges, concluding with the need for incremental innovation and pragmatic timelines.
Download the presentation
Various EU-funded R&I initiatives, members of the Cluster Hub, presented results and findings with the promise to reduce dependency on imports. RHINOCEROS project presented its intermediary results within the technical session “Recycling and circularity”, alongside sister projects RELiEF, RESPECT and FREE4LIB.
- Alvaro Manjon (TECNALIA) detailed the RHINOCEROS project’s progress in automating battery disassembly, optimising recycling routes and upscaling processes for high recovery rates of battery materials, with a focus on sustainability and safety. The project is transitioning from R&D to construction and commissioning of pilot plants, with detailed engineering completed. Upscaling activities focus on refining, pre-treatment, and qualification of recovered materials for use in new batteries, aiming for TRL 6 and production of tens of kilograms of electrode materials per day. RHINOCEROS integrates circularity and zero-waste strategies, with ongoing validation of recovered materials in prototype batteries.
Download RHINOCEROS presentation - Thomas Opsomer (ABEE) presented the final results of RELiEF, a project that achieved a reduction in hazardous elements, developed a microwave leaching process with over 95% yield, and produced high-purity lithium carbonate (98.28%); while not yet battery grade materials, recovered lithium carbonate showed promising results in relithiation tests. The lithium carbonate produced was successfully tested in coin and pouch cell formats, demonstrating high reproducibility and performance close to commercial LFP materials. Opsomer emphasised the growing importance of LFP batteries in Europe and the lack of recycling capacity, noting that extended producer responsibility (EPR) regulations now require battery integrators to address recycling, registration, and reporting. This creates both administrative burdens and incentives for recyclers, with recycling fees sometimes reaching 30% of battery unit costs.
Moreover, RELiEF’s “balance sheet” listed its unsolved challenges which include insufficient black mass refining capacity and the upscaling of innovative technologies. On the other hand, opportunities include standardising black mass quality, implementing direct recycling methods and the development of regional ecosystems that connect recyclers, OEMs and policymakers to promote sustainable business models.
Download RELiEF presentation - FREE4LIB, presented by Juan Castro Bustamante (CARTIF), combines robotic dismantling, material recovery with a digital battery passport for traceability. The project achieved pilot-scale robotic dismantling, developed advanced state-of-health estimation tools for second-life batteries and implemented multiple pre-treatment and recycling technologies, including pyrolysis, ultrasonic delamination and hydrothermal routes for cathode material recovery. Recovered metals and polymers were reused in new battery packs, including e-bike prototypes, with successful demonstration of material quality and performance. The project also established a pilot line for metal powder atomization and 3D printing of recovered plastics.
Project partners developed a digital passport featuring role-based access, blockchain integration, and QR labelling to enhance traceability and regulatory compliance. The platform will be made available for public access by the end of the project.
Download FREE4LIB presentation - RESPECT, represented by Andrea Locati (Chalmers University), presented the project’s developed flowsheet for solvent extraction and recovery of manganese, cobalt and nickel from black mass, achieving high purity and minimal cross-contamination. The recovered salts were used by CEA to produce cathode active materials, with up to 20% recycled content in electrodes, showing how the targets proposed by the Critical Raw Materials Act and the Battery Regulation are achievable. The approach allows for flexibility in product output, enabling adaptation to different NMC chemistries or other applications. Challenges include the complexity of operations, need for recirculation to minimise losses and adjusting process parameters to variable feed compositions.
The conclusions of the RESPECT presentation emphasised the need to propose clear definitions of battery-grade materials and the importance of aligning process outputs with regulatory requirements.
Download RESPECT presentation
Debating industrial integration, collaboration and European competitiveness
The second panel discussion, following technical session #2, moderated by Olga Henkele (Kellen) and inviting Joana Gouveia (INEGI), Sam Hoefman (EURICE) and Dr. Florian Anderhuber (Euromines) as guests, reflected on how Europe can progress from promising battery recycling innovations toward a more operational and industrial circular economy. Speakers agreed that while European research projects are generating strong technical evidence, several barriers still remain before a fully closed loop can be achieved. It addressed policy and market dynamics:
- Barriers to circularity: panelists emphasised the need for economically viable recycling in Europe and reducing regulatory burdens on recyclers. Other recommendations placed the time gap between research and industrial implementation, as well as cross-collaboration communication channels between academia, industry and policy makers as drivers of European industrial competitiveness.
- Florian Anderhuber highlighted lengthy permitting processes for new technologies and the lack of demand-side measures such as recycled content requirements. He stressed the need for financial support and market incentives to stimulate recycling and primary sourcing within Europe.
- Panellists discussed the importance of feeding clear evidence from EU-funded projects into policy making, setting clear project objectives, and considering the life cycle of projects from inception to exploitation. Joanna Gouveia, while emphasising on the LCA approaches in project, provided a direct example from the RELiEF project, where policy definitions limited the classification of secondary lithium, suggesting the need for broader definitions.
Read the entire review article
RHINOCEROS project validates extraction routes
The road to a green transition relies heavily on Europe’s capacity to electrify our way out of the use of coal, oil and natural gas before it becomes too late. A large part of this European vision counts on accelerating its transition to electric mobility and therefore upscaling its battery production. With scarce primary resources, EU policy makers push for the recycling of end-of-life [EoL] lithium-ion batteries [LIBs] as a strategic priority. More than recovering the metals trapped in defunct applications, the challenge lies in producing battery-grade materials that can reintegrate the supply chain. The R&I RHINOCEROS project has recently validated its extraction routes at laboratory scale, a preliminary step before pilot-scale implementation.
Electrochemical recovery of lithium
Researchers at Chemistry Department of Sapienza University of Rome [UoS] tested various electrochemical parameters to optimise Li extraction from the black masses [BM] obtained by the mechanical pre-treatment operations conducted in work package [WP4]. Their electrochemical process initially demonstrated its ability to extract Li without causing the dissolution of other cathode elements. Later, the research group replicated the electrochemical conditions in a two-chamber cell, where they achieved 82% Li recovery, later refined to produce LiOH with >99.5%.
Selective electrochemical recovery of Li using a two-chamber cell configuration. © University of Sapienza
Direct synthesis of cathode and anode materials via hydrometallurgical routes
Additional to electrochemical route, UoS also explored a hydrometallurgical route to synthesise Li-Mn-rich cathodes and reduced graphene oxide (rGO) directly from black mass. Applying Hummers’ method, researchers converted graphite into graphene oxide and co-precipitated metals to form Li-Mn-rich precursors. Electrochemical tests indicate that Li-Mn-rich cathodes derived from thermally treated black mass achieved performance comparable to those made from commercial salts, with capacities up to 202 mAh/g. Reduced graphene oxide synthesised from mechanically treated black mass displayed superior performance compared to rGO from commercial graphite.
Solvometallurgical recovery of Ni and Co
After studying the effects of the pre-treatment processes applied in WP4 to generate BM, researchers at TECNALIA [TEC] validated a solvometallurgical process using deep eutectic solvents (DES) to recover nickel and cobalt under mild conditions. The process achieved >95% metal leaching efficiency and precipitation yields purity.
Moreover, researchers have confirmed the scalability of the solvometallurgical process to large-lab testing without performance loss. To address the costs of this process, TEC researchers have also succeeded in demonstrating the reuse of DES up to 12 times across three systems.
Direct recovery of Ni/Co/Mn/Li with gas-diffusion electrocrystallisation
VITO advanced their proprietary gas-diffusion electrocrystallisation [GDEx] to recover Ni, Co, Mn and Li from leachates obtained from black mass and other WP5 partners. The process delivered >90% recovery rates for Ni, Mn and Co and>99% for Li in the form of layered double hydroxide (LDH) and spinel-type nanostructures for the synthesis of cathode active materials.
With better results obtained from the lithiated nickel manganese cobalt oxide (LNMCO) material synthesised from the leached black mass provided by ACC [thermal pre-treatment], VITO researchers validated its electrochemical activity as a cathode material for LIBs by assembling coin-type half cells. The recycled cathode material showed electrochemical activity, but the achieved capacity is lower than the expected which reveals the requirement of the optimisation process of upscaled extraction of metals, lithiation and slurry processing.
| Sample | Metal recovery (%) | |||
| Ni | Mn | Co | Li | |
| RHINO 2d | 90 | 89 | 96 | 99 |
Optimised recovery of materials from low concentration waste streams
A different task in the refining work package, led by LEITAT and TEC, aims at the recovery of low concentration materials from leachates and effluent streams produced in previous refining tasks. Researchers have developed polymer inclusion membranes (PIMs, LEITAT) and electrochemical systems (TEC) to recover metals from low-concentration streams, achieving up to 80 % recovery for Co and Mn, up to 60 % for Ni and >70 % lithium precipitation with high carbonate purity (>94 %).
In the case of the polymer inclusion membranes, several experiments were performed to define and select both the adequate extractant and the optimal operational conditions. Additionally, selectivity tests are being conducted with a focus on assessing the selectivity of PIM towards a specific metal. Finally, the selected PIMs were defined as follow:
| Series | Thickness | Polymer | Plasticizer | Carrier |
| Mn-PIM-1 | 30±2 um | CTA | 2-NPOE | DEHPA |
| Co-PIM-1 | 28±4 um | CTA | 2-NPOE | Cyanex 272+TBP |
| Ni-PIM-LIX 1 | 40±4 um | CTA | 2-NPOE | LIX84I |
Lab scale validation of most promising routes
After a multi-criteria assessment considering technical performance, eco-efficiency, and scalability, the TEC solvometallurgical route using ACC thermal black mass was selected for pilot-scale validation. This process demonstrated the best balance of recovery efficiency, cost, and environmental impact, and will serve as the reference flowsheet for WP6 upscaling.
Results evaluation at lab scale and prior to upscaling. ©TECNALIA
The RHINOCEROS project has reached a turning point in its efforts to automate the dismantling of end-of-life [EoL] electric vehicle batteries. If in the previous communications, researchers at the Faculty of Engineering and Science within the University of Agder [UiA] were developing digital simulators and algorithms to plan disassembly sequences, this reporting brings updates about the validation of the robotic systems and the introduction of a cognitive agent for adaptive execution.
Researchers first developed a Disassembly Process Plan [DPP], where they optimised complex scheduling problems, such as planning time and tool allocation for dismantling operations. More recently, the UiA team commissioned a dual robot cell in ROS2 simulation, step which allows engineers to validate motion planning, tool changes and coordination without risking costly errors.
After simulation, the robots and tools moved into lab trials and applied real-world settings. Standard off-the-shelf tools proved inadequate for disassembly operations. To address this, researchers have firstly upgraded tools with higher torque, better locking systems and simplified geometry that enable the robot to manage modules not only safer, but also faster. Redesigned tools already proved their capabilities for a variety of operations: unscrewing, cutting cables, lifting modules or changing tools.
Cognitive agent
The latest advancements brought by UiA include a cognitive agent that connects a digital battery repository with the robotic platform. The repository acts as a knowledge library that stores connection maps, 3D models, disassembly rules and historical data from previous operations. This integration works both ways: the agent reads from the repository and updates it with new knowledge retrieved during dismantling. This way, the database improves over time. Access to this database allows the agent to adapt to three scenarios:
- Known structures: deterministic optimisation using genetic algorithms and Proximal Policy Optimisation [PPO] – allowing the system to find efficient dismantling sequences without getting stuck in poor strategies.
- Partial knowledge: behaviour trees for local recovery.
- Unknown structures: learning-based strategies trained in NVIDIA Isaac simulation.
Beyond planning, the agent translates plans into commands that can be executed by the robot, closing the gap between decision-making and action. It also learns from failure: for instance, when a tool slips or a path is blocked, the agent adjusts its strategy through reinforcement learning, improving performance over time.
The cognitive agent reduces planning time by up to 20% and improves resilience under uncertain situations. It is ready to communicate with the robotic cell during the next development stages which aim to automate 75% of the dismantling operations.
Future developments include additional visual inspection and CAD reconstruction to support second-life battery applications, along with other features: enhanced learning, handling new battery designs and other complex products and industrial scalability.
The last consortium meeting included a session dedicated to ongoing clustering activities, where the RHINOCEROS partners invited the RESTORE project coordinator, Christophe Aucher, Area Manager Energy Storage at LEITAT. The objective of this initiative is to introduce the two EU-funded projects, both focusing on advancing battery material treatment and recycling technologies. Both projects are members of the Cluster Hub “Production of raw materials for batteries from European resources”.
The RESTORE project introduced its approach to treat black mass – from sorting to discharging, retrieving all available materials and validating them in a coin battery. This concept aligns closely with the philosophy of the RHINOCEROS project, emphasising the importance of contactless sorting and efficient battery discharge processes. The aim of RESTORE is to remove cables and discharge batteries within 40 minutes, which would bring a big step towards more efficient recycling processes.
During the event, representatives of both projects addressed various industry challenges. One major issue is the profitability of selling black mass to states outside of EU, compared to the domestic scenario for producing electrodes. Additionally, the European market faces a slowdown in battery production, showing citizens preference for hybrid cars over electric vehicles. Nevertheless, both sides agreed on the need for critical raw materials, indispensable to emerging industrial sectors, underscoring the importance of sustainable and efficient recycling processes.
The RESTORE project introduced several technological advancements, including the KYBURTZ cell-to-electrode direct recycling method developed by VITO. This method aims to streamline the recycling process and enhance the quality of recovered materials.
Although the two projects share similar objectives and consortium partners, technical questions were not missing, addressing mainly the electrochemical recovery of lithium, the impact of the aqueous media on the quality of retrieved material and the potential to work in batch mode.
This first online encounter of RHINOCEROS and RESTORE sparks a lot of curiosity, key ingredient of innovation and technological advancement. Prospects already show opportunities to stimulate collaborations and knowledge exchange in the battery recycling R&I field.
Researchers at the Faculty of Engineering and Science within the University of Agder [UiA] have developed a digital simulator along with an algorithm that automatically generates the Disassembly Process Plan [DPP] for batteries. The algorithm is built to select autonomously the most suitable machine to execute each disassembly operation, along with the corresponding toolkit. Beyond automating the DPP, this technological breakthrough promises to reduce the total disassembly process duration.
With the electrification of mobility, the upcoming wave of e-waste will be hard to deal with. In general terms, electrical waste is shredded in bulk before sorting and reprocessing. But lithium-ion batteries [LiBs], the types used in EVs, are inflammable and request careful handling. Moreover, shredding lots of different types of e-waste simultaneously inevitably results in contamination. Separating components before shredding would yield greater levels of purity, even allowing various components, such as cathodes, to be reused in their entirety. Dismantling batteries is a dangerous operation due to the risk of fire or explosion. Nonetheless, this process typically involves manual labour to remove the casing and separating the internal components – electrodes, electrolyte, cabling and separators.
Within the RHINOCEROS project, the UiA is responsible for developing an automated system for characterising battery state, discharging via the grid and dismantling for reuse or recycling. Their ultimate goal is to reduce the operational duration and to improve resource utilisation. An important phase of their work lies in the development of a Disassembly Process Plan – shortly DPP, which features a disassembly sequence plan [DSP] and an algorithm that can establish autonomously the most suitable equipment for each disassembly operation. To avoid several rounds of unsuccessful trials, researchers have firstly created a digital simulator where they already tested the algorithm generating the DPP.
Trained to use time and resources efficiently
The use of a simulation environment offers a safe and cost-effective way for researchers to test and refine the algorithm in a controlled context. Beyond safety and resource optimisation, simulators facilitate also scalability and reproducibility. The algorithm is trained to select the most suitable toolkit and calculate optimal tool change sequences, which reduces the overall disassembly process duration. Moreover, it also features integrated data that allow it to verify beforehand compatibilities between requested tools and machine capabilities. By determining optimal disassembly sequences and tool allocation, the DPP reduces operational costs through more efficient equipment usage.
Closer look at the technical specifications of the DPP developed in RHINOCEROS
From a technical point of view, the digital simulator, built on a foundation of Python and NumPy, tracks job progress, machine availability, tool states and temporal dependencies. Researchers applied various Reinforcement Learning (RL) algorithms, including Proximal Policy Optimisation (PPO), Policy Gradient (PG), Advantage Actor-Critic (A2C) and Asynchronous Advantage Actor-Critic (A3C), to solve the Job Shop Scheduling Problem (JSSP). The PPO algorithm, in particular, has demonstrated superior performance compared to other algorithms and state-of-the-art (SoA) solutions. The simulator is using datasets similar to the battery disassembling problem, mimicking real use case scenarios.
In a recent development, researchers have introduced an innovative approach called QTM (Q-learning with Tsetlin Machines) to improve scheduling algorithms. This new method combines the pattern recognition abilities of Tsetlin Machines (TM) with the decision-making strengths of Q-learning. The QTM approach uses a sophisticated reward system to balance the completion of tasks, minimize the time taken, and optimise the overall schedule. The TM component excels at identifying patterns in scheduling scenarios by analysing key features of jobs and operations. Meanwhile, the Q-learning framework provides a foundation for learning through a process of trial and error, gradually improving decisions over time.
The DPP developed in the RHINOCEROS project features PPO, QTM and classic deep Q-learning.
Impact on the real-world industrial applications
The DPP brings significant implications for real-world applications, particularly in industries that prioritise efficiency and sustainability. In EV battery recycling, it optimises the disassembly sequence, enhancing efficiency. It is also applicable to electronics WEEE recycling, where complex assemblies require precise disassembly. The DPP can integrate into large industrial scales, enabling the reuse of valuable components and supporting circular business models.
Automation integrators and robotic system developers benefit from the DPP’s computational framework, which models tool-changing operations and associated costs, optimising robotic movements and tool selection. Academic institutions can use the DPP to study broader questions in automated planning and robotic intelligence, providing a well-defined problem structure for testing new algorithms and heuristics.
For industrial research labs, the DPP offers a framework to develop specialised applications across different product categories, evaluate alternative product designs for end-of-life processing efficiency and simulate different disassembly strategies before physical implementation, reducing development costs and accelerating innovation cycles in recycling technologies.
Future developments for the Disassembly Process Plan
Plans for the DPP foresee its integration into a comprehensive digital twin of the manufacturing environment. Before its demonstration in a real-world setup, this digital twin operating with configurable tool capabilities and interdependent job sequences, will have to pass thorough testing in a simulated environment.
© Visual: University of Agder [UiA]
Lithium-ion batteries proved to be one of the most efficient energy storage solutions, widely used for applications such as electric vehicles [EVs] and renewables. Despite faltering economies and material shortages, more than 40 million EVs are expected on the EU’s roads by 2030. With mass electrification of transport, there will be an important demand for batteries and the materials they are made from. Despite an incentivising regulatory context at EU level, in 2023 Europe was home to only 1% of the production of key battery raw materials. European carmakers and battery cell producers are already fret about volatile prices and limited supplies of cathode and anode materials. But with EV fleet number growing on the EU’s roads, the volumes of End-of-life [EoL] batteries will considerably increase in the near future. And so will the pressure to recycle closer to home, recovering key components and mitigating the supply chain risks.
Conventional recycling methods often require high consumption of energy and reagents. But electrochemistry offers a promising green alternative method, enabling selective recovery of Li without the need for chemical extracting compounds.
Selective electrochemical extraction of lithium
Researchers at Chemistry Department of Sapienza University of Rome [UoS] tested various electrochemical parameters to optimise Li extraction from both commercial cathode material and the black masses [BM] obtained by the mechanical pre-treatment operations conducted in work package [WP] 4.
The electrochemical tests on high-purity commercial cathode materials [LiMn2O4, LiCoO2 and LiNi1/3Mn1/3Co1/3O2] allowed a selective extraction of Li up to 98 %. UoS researchers later applied the same electrochemical process on the black mass recovered by the mechanical pre-treatment, obtaining a lower Li extraction yield of 82 %. This decreased yield was mainly attributed to the presence of conductive carbon which undergoes a simultaneous electrochemical oxidation. Analysis of the resulting electrolyte at the end of delithiation experiments showed negligible extraction of cobalt (Co), nickel (Ni), and manganese (Mn). This demonstrated the method’s ability to selectively extract Li without causing the dissolution of other cathode elements.
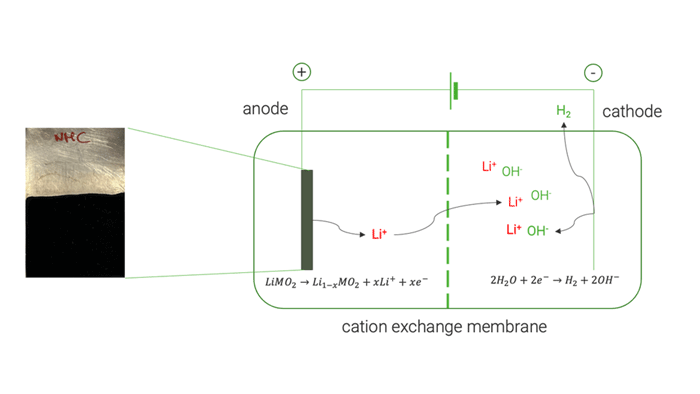
Cation exchange membrane | © Univ. of Sapienza
The optimised electrochemical conditions applied on the BM were replicated in a two-chamber cell to facilitate the separation and concentration of extracted Li on the cathode side, achieving approximately 85 %. The resulting solution was used to crystallise LiOH, resulting in a mixture of LiOH and LiOH·H₂O, with a purity exceeding 99.5 %.
Synthesis of reduced graphene oxide (rGO) and Li-Mn rich cathode material
The direct synthesis of high-value products from EoL LIBs, bypassing the complex and expensive separation of different metals, can be achieved through a competitive recycling strategy. The simultaneous synthesis of reduced graphene oxide (rGO) and lithium-manganese-rich (Li1.2Mn0.55Ni0.15Co0.1O2 – LMR) cathode material from EoL LIBs represents a promising approach to enhance the economic feasibility of hydrometallurgical recycling processes by producing high-value-added advanced materials.
UoS researchers developed an innovative recycling process to directly synthesise a layered LMR cathode and rGO from EoL LIBs. The proposed recycling strategy relies on the application of Hummers’ method to process the electrodic powder delivered by pilot-scale mechanical pre-treatment of mixed Li-ion batteries. The Hummers’ method has allowed for the quantitative extraction of metals from the electrodic powder and the production of GO without any metal impurity. The resulting solution contains the metals from the cathode materials (Co, Ni, Mn) with a significant amount of manganese from the KMnO4 used in the Hummers’ method. From such consideration stems the idea to synthesise LMR cathode material.
UoS researchers synthesised GO using the black masses obtained from three distinct EoL LIB pretreatments performed in WP4. The effects of thermal, mechanical and mechanochemical pretreatments were evaluated using the widely adopted Hummers’ method for graphene production. The study conducted by UoS researchers examined both pristine black masses and those subjected to metal removal via conventional acid leaching, a process frequently employed in LIB recycling to extract metals from cathode materials. This metal removal procedure had a notable effect on introducing oxygen functional group defects across all samples. A synergistic effect was particularly evident in the mechanochemically treated black mass, where acid leaching increased the GO yield significantly. This remarkably high graphite conversion to GO underscores the importance of selecting appropriate pretreatment methods for EoL LIBs, particularly when aiming to integrate advanced materials like GO into the recycling process.
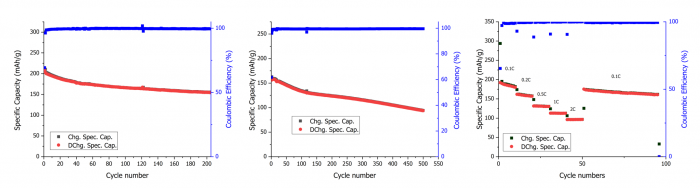
Cycling performance of LMR synthesised from thermally treated black mass: (a) Galvanostatic cycling at 0.1C, (b) Galvanostatic cycling at 1C, and (c) Rate capability performance with the current increasing from 0.1C to 2C every 10 cycles | © UoS
Recovery of Ni/Co materials by solvometallurgical route
RHINOCEROS is a research project that aims to o create economically and environmentally sustainable methods for reusing and recycling LIBs. The project focuses on developing cost-efficient, flexible and eco-friendly processes to recycle all materials in LIBs, including metals, graphite, fluorinated compounds and polymers.
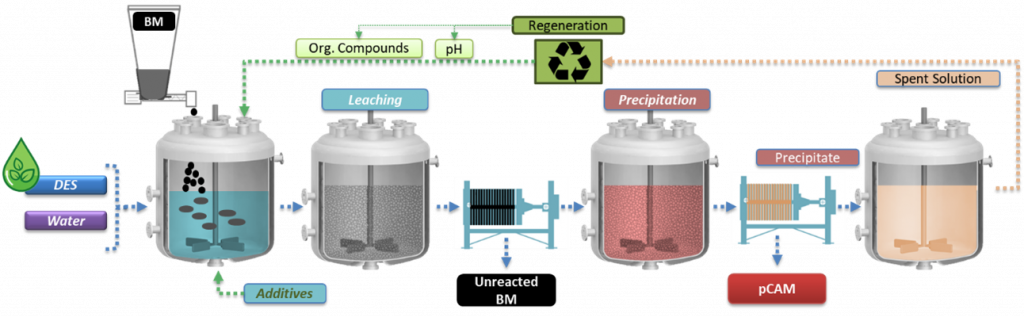
Flowsheet of the leaching, precipitation and regeneration process | © TEC
After studying the effects of the pre-treatment processes applied in WP4 to generate BM, researchers at TECNALIA [TEC] tested various solvometallurgical routes to extract critical metals like Ni, Mn and Co from LIBs under mild conditions. They established optimal process conditions to achieve leaching and precipitation efficiency along with high purity of the produced precursor of cathode active materials (pCAM) under mild conditions. This approach also enhances the recyclability of the extractant.
After characterising all the input materials received from the pretreatment operations, TEC research group analysed also the obtained leachates. The leaching process was optimised at laboratory scale by studying the effects of various parameters such as leaching time and temperature, stirring, waste/liquid ratio and additives.
The solvometallurgical process for recycling end-of-life [EoL] battery BM has successfully developed high-yield, selective systems, validated across various NMC chemistries and black mass pretreatments, achieving over 95 % leaching efficiency. Controlled precipitation delivered up to 95 % precipitation efficiency, producing precursors with more than 99 % purity, meeting project targets. Moreover, researchers have confirmed the scalability of the solvometallurgical process to large-lab testing without performance loss. To address the costs of this process, TEC researchers have also investigated alternatives to reuse and recycle the deep eutectic solvent [DES] leaching spent solution, reporting its dependence on the properties of the BM feed.
The residue after leaching – the graphitic anode material – has been purified via hydrometallurgical routes, underscoring the difficulty of removing the most refractory phases resistant to harsh leaching conditions.
Optimised recovery of materials from low concentration waste streams
A different task in the refining work package, led by LEITAT and TEC, aims at the recovery of low concentration materials from leachates and effluent streams produced in previous refining tasks. It is divided in three subtasks, each exploring different membrane-based technologies to achieve a zero-waste strategy for recovering remaining elements.
The objective of the first subtask is to develop new extractants based on ionic liquids or DESs, to be used within novel Polymer Inclusion Membranes (PIMs), for the recovery of Li from solvometallurgical refining final streams. The team firstly analysed the SoA on Li extraction strategies, selected, synthesised and characterised different potential extractants based on hydrophobic deep eutectic solvents (HDESs). Selected HDESs were synthesised and sent to the next subtask devoted to PIM fabrication and testing using different ILs and/or synthesized HDESs. These membranes represent an innovative technology that effectively adapts to the specificities of each input stream, enabling optimal recovery of valuable metals present in the waste generated by processes carried out. In the this subtask, the PIMs yielded the following recovery rates:
- Co and Mn: efficiencies close to 80 %
- Ni: 60 % recovery
- Li: only 19 %, but exhibiting a high flux rate of 1.2 mol/m²·h, suggesting potential for larger lithium recovery.
Finally, in the last subtask, researchers developed a three-chamber electrochemical process for the extraction, concentration and recovery of lithium in the form of lithium carbonate from solvometallurgical final streams. Different ion-exchange membranes were characterised and lithium carbonate precipitation step was studied and optimised. They tuned the overall process conditions achieving more than 70 % Li extraction from leachate to catholyte and high lithium carbonate precipitation yields (>70 %) with high purity (>94 %).

Three-chamber electrochemical cell | © TEC
Extraction of Ni/Co/Mn/Li with GDEx
Gas-diffusion electrocrystallisation [GDEx] is an innovative electrochemical process used to recover metals from complex fluids, such as leachates, industrial waste streams and organic solutions. This innovative process has been successfully demonstrated in multiple research projects, proving its capability to recover valuable metals while operating under low-cost and environmentally friendly operating conditions. The GDEx process has achieved Technology Readiness Level [TRL] 3 for the recovery of Co and Mn from synthetic solutions and attained TRL 4 in treating black mass leachates. One of the multiple advantages of this electrochemical process is its capacity to recover Ni, Co and Mn simultaneously, with the flexibility to manipulate operational conditions to obtain targeted products.
Specifically for the battery recycling sector, the GDEx process offers various advantages over conventional battery recycling methods:
- High recovery efficiencies, exceeding 90 % for Co and Mn and over 90 % for Li, with potential for further optimisation.
- Low energy consumption: less than 20 kWh per kg of recovered material, which reduces the operational costs significantly.
- Minimal chemical input: the process relies primarily on air and in-situ synthesised oxidising/reducing agents, ensuring high material efficiency and circular use of non-hazardous chemicals.
- Mild emissions, with the possibility to further treat any effluents using conventional wastewater treatment methods.
- Low temperature operation: from room-to-mild temperatures [18 – 70 °C].
- Direct synthesis of functional battery materials, such as Co/Ni/Mn spinels and birnessites, which can be used to manufacture new lithium-ion battery electrode materials and eventually LIBs. This feature eliminates the need for additional refining steps, streamlining the production of new batteries from recycled material.
Within the RHINOCEROS project framework, researchers at VITO have been finetuning the operational parameters of their proprietary GDEx process, additionally testing and validating the recovered materials through the synthesis of high-performance electrodes for next generations batteries, demonstrating therefore the circularity, cost, environmental and social benefits of the solutions developed.
After testing the direct recovery route of Co, Ni, Mn and Li on synthetic solutions with the main objective to finetune the GDEx operating settings, VITO researchers have been implementing the process to the BM samples provided by the pre-treatment operations [ACC and TES] and to the aqueous extractants processed by the hydrometallurgical route explored by UoS and the solvometallurgical route explored by TEC. Additionally, they also documented the testing operations using a 3D phase diagram material library, mapping the formation of different materials based on Co/Ni/Mn concentration ratios from synthetic solutions.
The GDEx process applied on the leachate solution obtained from the BM provided by ACC achieved high recovery rates: 90 % for Ni, 89 % for Mn and 96 % for Co.
On the other hand, GDEx process implemented on the leachate solutions provided by UoS [originally from the BM of TES] results in the metal extraction efficiencies of 98 % for Ni, 68 % for Mn, and 96 % for Co. Sequential GDEx experiments have potential to improve the recovery efficiency by removing the impurities first and then the recovering the targeted metals. GDEx treatment on TEC’s leachate solution required optimisation due to the presence of high content of Cu and Al impurities.
With better results obtained on the lithiated nickel manganese cobalt oxide (LNMCO) material synthesised from the leached black mass provided by ACC [thermal pre-treatment], VITO researchers validated its electrochemical activity as a cathode material for LIBs with coin-type half cells. The recycled cathode material maintained a stable charge-discharge profile up to 50 cycles, with an initial charge-specific capacity of 37 mAh g⁻¹ and a reversible capacity of 26 mAh g⁻¹. Later, a batch with large amount of BM will be treated by GDEx process to optimise the electrochemical performance of the recycled electrode material.
After removing the targeted transition metals Ni, Co and Mn from the leachate solutions of the BMs, researchers used the same solution to extract lithium, recovering >99 % Li from the leachate of ACC BM.
The low-temperature or near-ambient operation condition of GDEx process eliminates CO₂ emissions, ensuring a clean and cost-effective approach to recovery of NMC precursor. This aligns with RHINOCEROS project goals, promoting a circular and sustainable battery materials economy. These results highlight the sustainability and scalability of the GDEx process, providing a carbon-free alternative for battery material production, aligning with global efforts to decarbonize critical raw material supply chains.
© visual: VITO
The challenge of recycling lithium-ion batteries
Current battery recycling is confined to recovering raw materials from the scrap produced by gigafactories. But with the adoption of lithium-ion batteries (LIBs) increasing, a much richer vein will soon emerge, as the first wave of electric vehicles (EVs) reach the end of their lives. And with it, an additional challenge emerges managing the growing waste generated by these energy storage applications. Traditional recycling methods, such as pyrometallurgy and hydrometallurgy, have faced certain hurdles in efficiently recovering valuable materials, mainly due to the presence of the organic components present in the battery binders and separators. Some methods are labour-intensive, whereas others need lots of energy or are environmentally harmful. But each hurdle is another opportunity for innovation.
Optimisation of shredding pre-treatment: TES develops and validates their mechanical process for battery recycling
Leveraging over 20 years’ experience in battery recycling, TES partners oversee the development of a shredding process for batteries using its patented in-house technology. Over the first two years of the RHINOCEROS project, they have been conducting various activities using commercial battery packs, from evaluation and deactivation to pack-to-cell dismantling.
TES have been prioritising safety measures and efficiency, ensuring the system extracts the residual energy stored in batteries, preventing it from going to waste. Following the discharge process, TES applies its proprietary mechano-physical pre-treatment, which involves pre-shredding under an inert atmosphere. This is followed by physical separation techniques to recover polymers, base metals (Al, Cu, Fe), and black mass containing valuable active materials such as Co, Ni, Mn, Li and graphite.
Despite the effectiveness of the current mechano-physical treatment, TES has identified areas for improvement. Moving beyond the state-of-the-art [SoA], TES has optimised its process with a pre-drying step to minimise solvent residues and enhance delamination and separation efficiency. These advancements have led to significant improvements in material recovery and product quality, making the recovered black mass more suitable for downstream hydrometallurgical processing.
- Electrolyte recovery rate: exceeding 95 %
- Polymer recovery rate: exceeding 80 %
- Lithium recovery rate: surpassing 95 %, with the majority retained in the black mass
- Impurity levels of Cu and Al: reduced below 1 %
Optimisation of thermal pre-treatment process
However, reintegration of cathodic and anodic materials into the production chain of new batteries remains a challenge. Thermal pre-treatment of the batteries remains paramount in deactivating, releasing and separating the battery materials. But before reaching the thermal pre-treatment, batteries undergo a sorting process according to the cell type. Discharging is not mandatory prior to the pre-treatment.
ACCUREC [ACC] partners have been modifying their thermal pre-treatment process, including a preceding stage for cell solvent recovery, which places their process beyond the SoA. To explore this technological path, they experimented various parameters altering the working temperature and the pressure of solvent distillation, to address safety concerns and stabilise the background electrolyte. A selective recovery of solvent was possible under specific temperature and pressure conditions achieving high purities >99.6 %. However, additional refining steps are necessary prior to further use in battery manufacturing. ACC partners managed to deactivate batteries completely and produce high-purity black mass [BM] with recovery yields of >95 %, using high temperature pyrolysis and a two-stages screening process.
Their process demonstrates that the distillation process prior to the thermal treatment allows the extraction of solvents used in the cell and streamlines high recovery rates of active material. However, their research concludes that the processing of end-of-life [EoL] batteries remains a trade-off between recycling efficiency and purity of the obtained fractions due to the large number of compounds and lack of control over the starting material.
Electrolyte recycling
The safe and environmentally responsible recovery of electrolytes is crucial in the LIBs recycling process. The SoA non-aqueous liquid electrolytes consist of a conductive salt (lithium hexafluorophosphate, LiPF6) dissolved in a mixture of organic solvents and additives. Many of these components are highly volatile, flammable, and classified as toxic. The current state-of-the-art LiB recycling process begins with the production of black mass, obtained through a series of pre-treatment steps. But uncontrolled evaporation of volatile electrolyte solvents during these stages presents major safety hazards. The research group at Technical University of Chalmers [CHA] have been exploring various processing pressure and temperature parameter settings for their supercritical carbon dioxide [scCO2], with a particular focus on the extraction of ethylene carbonate [EC].
CHA researchers successfully developed and validated the scCO2 process for electrolyte recycling from LIB black mass. The results demonstrated a >95 % overall recovery, with individual yields of >99 % for DMC, EMC, and DEC and 95 % for EC, using specific process conditions. The recovered EC retains its original structural integrity, confirming its chemical stability and purity.
But beyond the high purity and selectivity of carbonates, the developed scCO2 does not require any chemical reagents. Moreover, the used CO2 can be indefinitely recycled within the system, turning it into a clean separation process that eliminates toxic gas during processing.
Reactive milling for black mass production
The research group at KIT received black mass samples from partners ACC and TES. Only the samples from TES were fit to study the mechanochemical transformation of BM, as the ACC supply was already in a reduced state. The BM supplied by TES consists mostly of NMC (lithium nickel manganese cobalt oxides) cathode material and graphite, which was found to slow down the reaction kinetics.
KIT researchers applied their reactive milling on the BM provided by TES to reduce the cathode material to its metallic form and produce water-soluble lithium salts. They optimised the process using three types of mills [two planetary mills of different sizes and a SPEX shaker mill], engaging three types of reagents: Al, Mg and Ca. They also investigated the influence of milling parameters on the process duration and identified the most optimal parameters for faster kinetics. The planetary mill showed better results than the shaker mill for Mg and Al, while Ca performed well in the SPEX mill.
In depth studies performed by KIT concluded that the process is a mechanically induced self-propagating reaction, which is initiated by mechanical energy, such as the impact energy from the milling balls. Once started, it propagates through the material on its own, similar to a chain reaction. This type of reaction is highly exothermic, releasing a significant amount of heat, which further drives the reaction forward. Small amounts of volatile compounds, such as residual solvents or moisture, were found to have a significant effect on the reaction time. The basis of the volatiles interference is not clear yet, but it seems to be important to remove these compounds before the milling process to ensure a more efficient and faster reaction.
Further experiments are needed to scale up the process and ensure that it can be applied to larger batches.
Vacuum pyrolysis and Supercritical CO2 [scCO2] processing for binders recovery
Binders such as polyvinylidene fluoride [PVDF] and polytetrafluoroethylene [PTFE] for cathodes, and carboxymethyl cellulose [CMC] and styrene-butadiene rubber [SBR] for anodes, are particularly difficult to separate during mechanical processing. This reduces the efficiency of metal recovery and can lead to the formation of environmentally harmful substances, such as per- and polyfluoroalkyl substances [PFAS].
Thermal treatment methods, while effective in removing these organics, generate hazardous emissions like hydrogen fluoride [HF] and phosphoryl fluoride [POF3], necessitating stringent gas treatment measures. Additionally, battery separators, often composed of complex polymers like polyethylene [PE] and polypropylene (PP), pose further challenges due to their low economic value and the presence of residual hazardous materials.
ACC has optimised vacuum pyrolysis at 550°C, which enabled the complete decomposition of PVDF, and thus, the effective removal of binders and separators from LIB waste, while recovering fluorinated compounds from decomposition products. Generated HF was captured using two different methods: through potassium hydroxide [KOH] washing (producing potassium fluoride [KF]) or a calcite filter (producing fluorite).
Supercritical carbon dioxide [scCO₂] process technology has emerged as a promising alternative for LIB recycling, offering properties that are adjustable to organic compounds. However, PVDF dissolution in scCO₂ alone requires extreme conditions, making industrial implementation challenging. Chalmers researchers have explored possible co-solvent scCO₂ systems to enable the recycling of binder directly from mechanically treated BM. They developed a specialised extraction process using scCO₂ with a co-solvent (dimethyl sulfoxide, DMSO) to recover the PVDF binder from industrial battery waste. The process achieved a 55.6 % recovery rate of PVDF under mild conditions.
The scCO₂ technology was also applied to electrolyte and separator recycling, achieving over 90 % separator recovery with high purity.
© visual: ImageFlow via Adobe Stock
On 12 December 2024, the third edition of the annual workshop of the Cluster Hub “Production of Raw Materials for Batteries from European Resources” took place in Brussels, being co-organised by EU-funded projects RHINOCEROS, CRM-geothermal and CICERO. This third edition, along with an increasing number membership, confirm the hub’s role as a dynamic ecosystem that continues to generate innovations in the European battery materials sector.
The hub’s annual workshop, held as a satellite event of the Raw Materials Week 2024, provided once again a platform for presenting the most promising results from participating projects. Two technical sessions covered the entire battery value chain, from raw materials mining to recycling, while the opening conveniently portrayed the policy, the regulatory and strategic frameworks that support and drive the EU R&I initiatives in the battery sector.
Policy perspectives and supporting mechanisms for the battery sector
Susana Xara, Project adviser on raw materials at European Health and Digital Executive Agency (HaDEA), established the discussions tone, navigating through the insights of the Critical Raw Materials Act [CRMA] and the Net Zero Industry Act [NZIA] and focusing on their contribution to securing a sustainable supply of critical raw materials for the European battery industry.
Wouter IJzermans, BEPA Executive Director, presented the long-term vision and potential revisions of their roadmap, emphasising the importance of policy frameworks and incentives in promoting battery innovation and deployment across Europe.
The presentation of Vasileios Rizos from the Centre for European Policy Studies (CEPS) identified various barriers and challenges emerging from the EU policy framework on batteries, based on inputs from 20 companies across the entire battery value chain, including partners from the BATRAW project, member of the Cluster Hub since 2022. The representative of CEPS concluded with a set of policy messages referring to early dialogue channels established between policy-makers and various stakeholders. Before the legal requirements entry into force, this information exchange on availability of secondary data sets could enable stakeholders to assess the data quality, select suitable sets of information and identify potential data gaps.
Publicly available resources submitted by CEPS:
- Barriers and policy challenges in developing circularity approaches in the EU battery sector: an assessment – CEPS
- Implementing the EU digital battery passport – CEPS
- Compliance with the EU’s carbon footprint requirements for electric vehicle batteries – CEPS
Orchestrating the launch and on-going work of the Cluster Hub, PNO Innovation Belgium [part of PNO Group – leader in innovation and funding consultancy], represented by Dr. Nader Akil, concluded the first session with an overview of all EU funding programmes supporting research, innovation and investment in raw materials production for batteries. Additional to the upcoming funding opportunities and guidance on selecting the appropriate funding opportunities based on the status of technology, Dr. Nader Akil introduced another initiative launched by PNO Group – DIAMONDS4IF. This project supports the preparation of Innovation Fund applications, enabling the transfer of H2020 research results into successful ventures and securing investment funding.
Download Funding Schemes presentation
The third session of presentations commenced with an outline of the main findings of the LIFE DRONE project, which concluded in June 2024. Presented by Lorenzo Toro, process engineer at Eco Recycling, the project demonstrated the feasibility of producing high-quality NMC oxide and graphite from recycled batteries. The innovative process confirmed significant environmental benefits, with a reduction of 59 % in terms of kg CO2 eq. Additionally, one plant is estimated to treat 500 tonnes of batteries/year. The technical-economic evaluation of this industrial plant, with a potential capacity of 500 tons/year, showed a return on investment (ROI) of 31.64 % and a payback time (PBT) of 3.16 years. The analysis indicated that attractive payback times could be obtained even with varying prices for NMC and graphite.
Download LIFE DRONE presentation
RHINOCEROS presenting results of the Electrochemical Li recovery strategy from LIBs black mass
The electrochemical Li recovery from Li-ion battery black mass, investigated in the RHINOCEROS project and presented by Prof. Pier Giorgio Schiavi from Univ. of Sapienza reported Li extraction yields from end-of-life (EoL) LIBs in the range of 82 %, and faradaic efficiency compared to commercial cathode materials (close to 100 %). Researchers have investigated potential causes that could explain the relatively low selectivity ranges of Li and other metals available in black mass. The simultaneous oxidation of impurities found in the black mass can be a justified explanation for the preliminary results obtained. Experimental results show that the extraction percentages for Co, Ni, and Mn remain in very low ranges. When contemplating upscaling scenarios, Prof. Schiavi mentioned the researchers are evaluating an alternative approach that enables the treatment of larger quantities of powder without replicating the manufacturing process of LIB electrodes.
Download RHINOCEROS presentation
The session featured also presentations of other initiatives addressing the batteries recycling topic: Benjamin P. Wilson, Senior Scientist, Hydrometallurgy and Corrosion at Aalto University on behalf of the RESPECT project, Miguel Aguilar, researcher at LEITAT Technological Centre for the BATRAW projects, and Joana Gouveia [Researcher at the Institute of Mechanical Engineering and Industrial Management (INEGI) and America Quinteros [Researcher at LUT Univ.] for the ReLiEF initiative.
